EVIDENCE-BASED
INNOVATION
EVIDENCE-BASED
INNOVATION
Advancing coronary intervention procedures with innovation demands evidence. Elixir Medical technologies are supported with a growing body of clinical data. These global, multi-center studies demonstrate the safety, performance, and long-term patient outcomes across a diverse range of patient populations. Results have been peer-reviewed and published or presented at leading conferences, including TCT, EuroPCR and ESC.

Randomized
controlled trials

Real-world
registries

Studies confirming results with
OCT & IVUS imaging
DynamX® results across studies reinforce sustained treatment benefit
DynamX Coronary Bioadaptor System has been evaluated for acute performance, long-term durability, and restoration of function.
Studies have included head-to-head comparisons with a third-generation DES and real-world patients, including treatment in LAD lesions and patients with acute coronary syndrome (ACS).
- 445-patient RCT (1:1), multi-site compared to DES, data to 4 years
- 2,400-patient nationwide RCT (1:1) compared to DES, data to 2 years
Results from BIOADAPTOR RCT at 4 Years
Showing % of patients experiencing Target Lesion Failure (TLF) for bioadaptor vs. DES
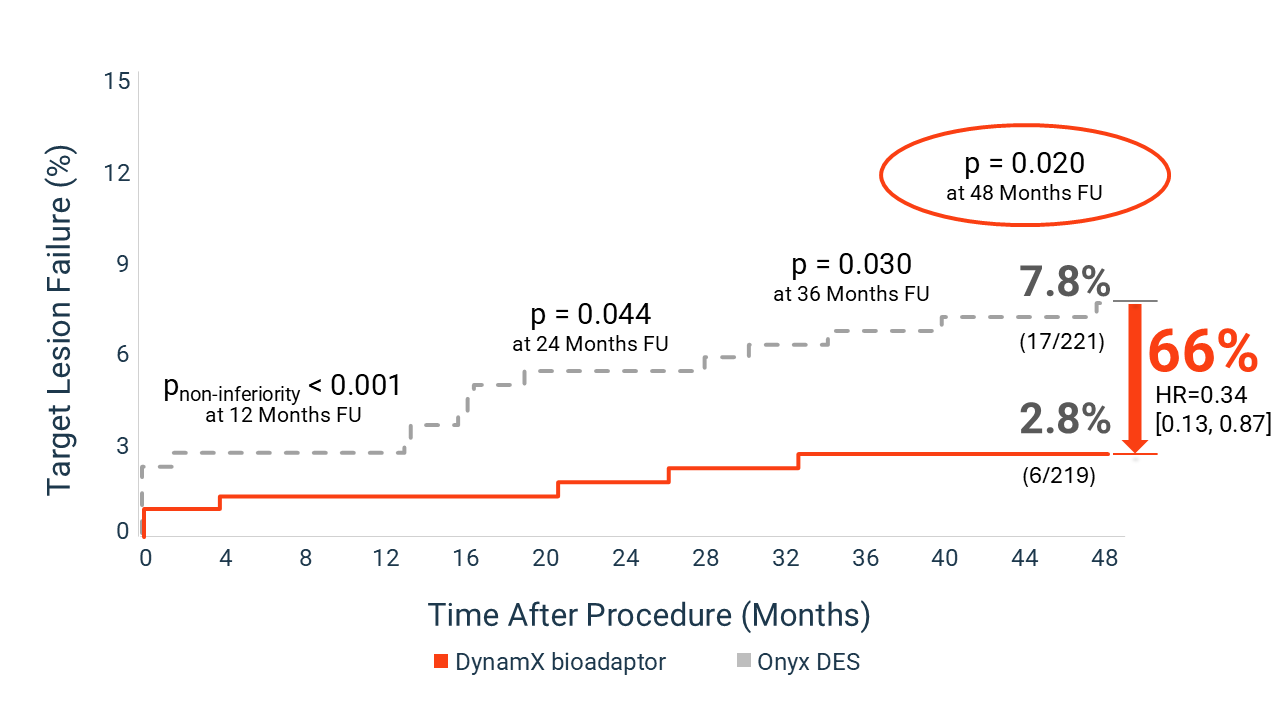
These findings support a treatment approach that focuses not only on vessel patency, but also on restoring natural vessel function and long-term vascular health.
BIOADAPTOR RCT
n=445
An RCT evaluating the acute and long-term safety and effectiveness of the DynamX Coronary Bioadaptor compared to contemporary drug-eluting stents.
The study showed:
- 1.8% TLF at 12 months vs. 2.8% for DES
- 16% increase in blood flow
- -9% volume regression in lipid-rich lesions vs. +10% for DES
- 66% reduction in TLF at 4 years vs. DES
INFINITY-SWEDEHEART RCT
n=2,400
A large-scale, real-world registry assessing DynamX Coronary Bioadaptor performance compared to DES in routine clinical practice.
Results demonstrated:
- 2.35% TLF at 12 months vs. 2.77% for DES
- 48% reduction in TLF vs. DES
- 57% reduction in TLF in ACS patients vs. DES
LithiX™ studies demonstrate versatility, safety and efficacy
LithiX Hertz Contact Intravascular Lithotripsy (HC-IVL) is supported by clinical research.
Confirmed with angiographic and intravascular imaging, these studies highlight simplified calcium modification with procedural efficiency and safety in complex coronary lesions.
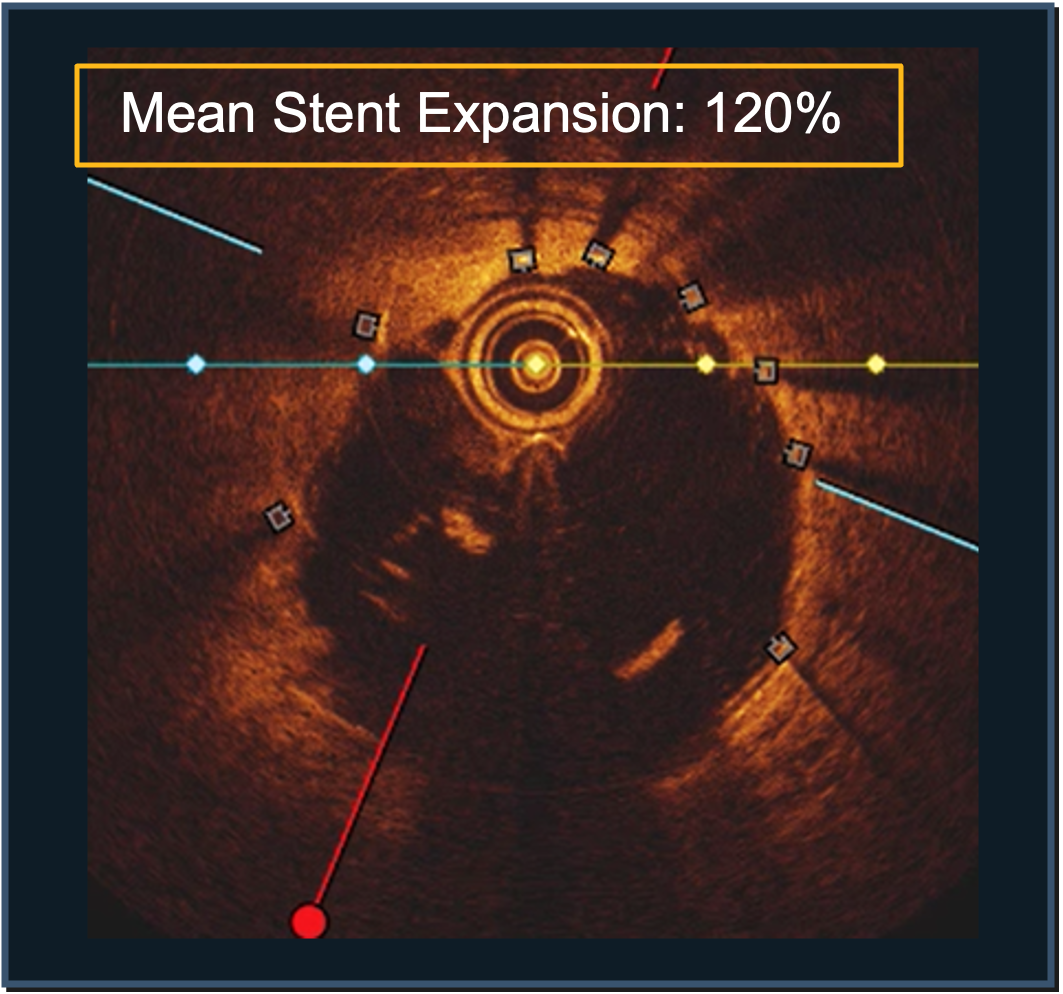
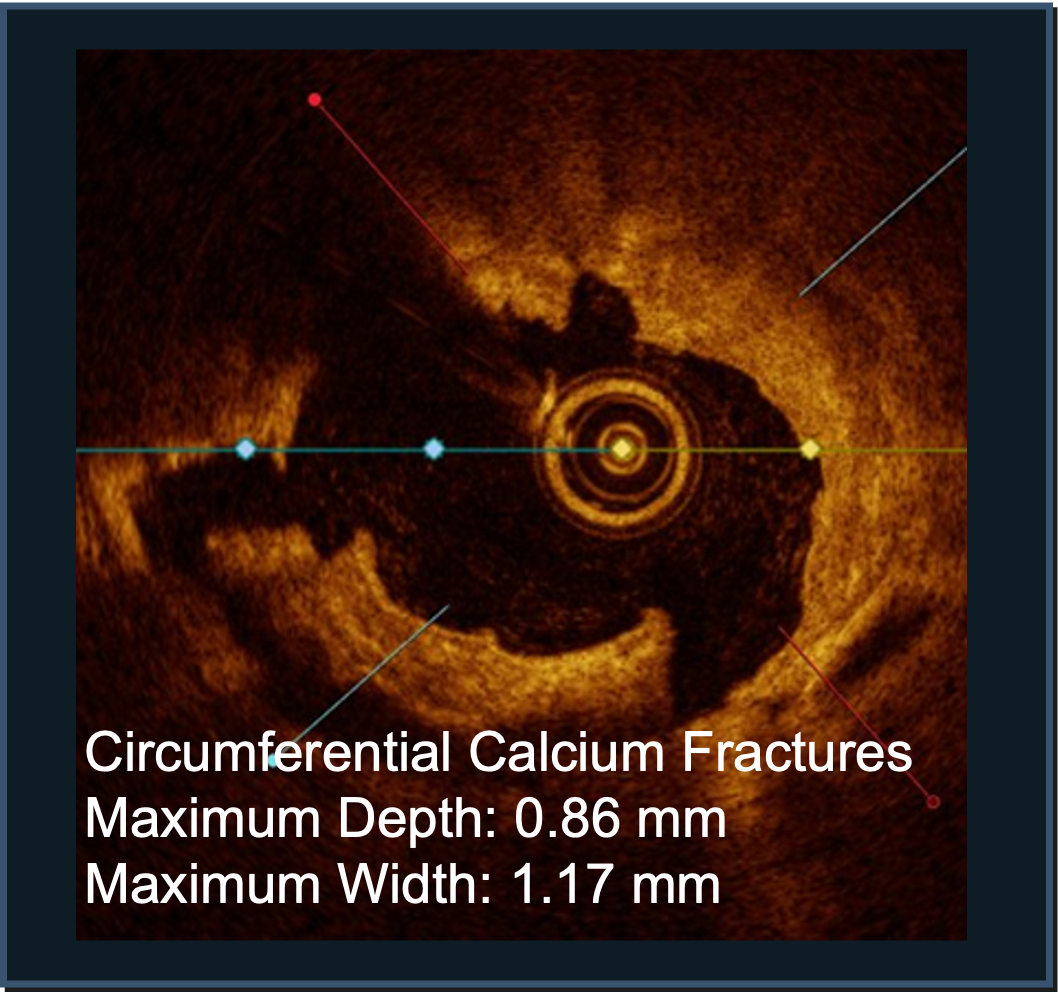
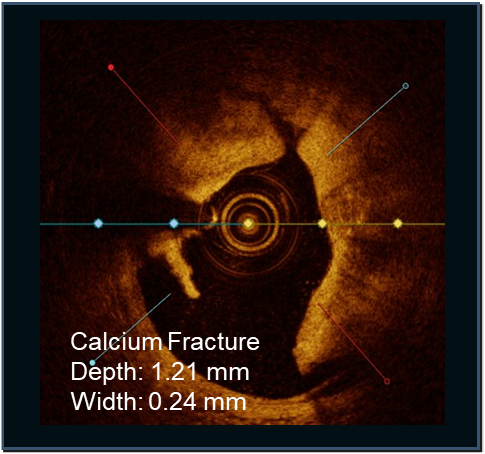
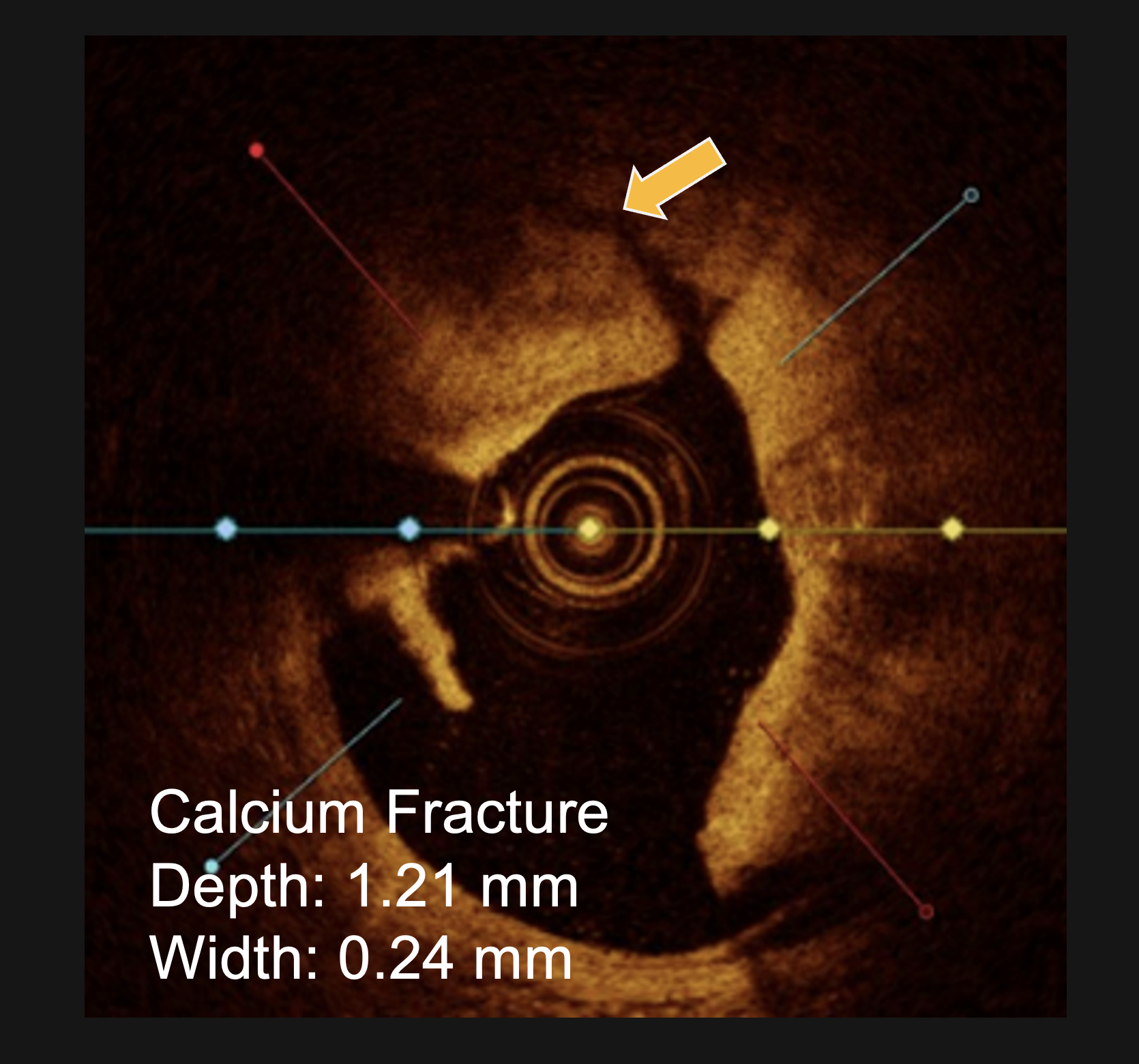
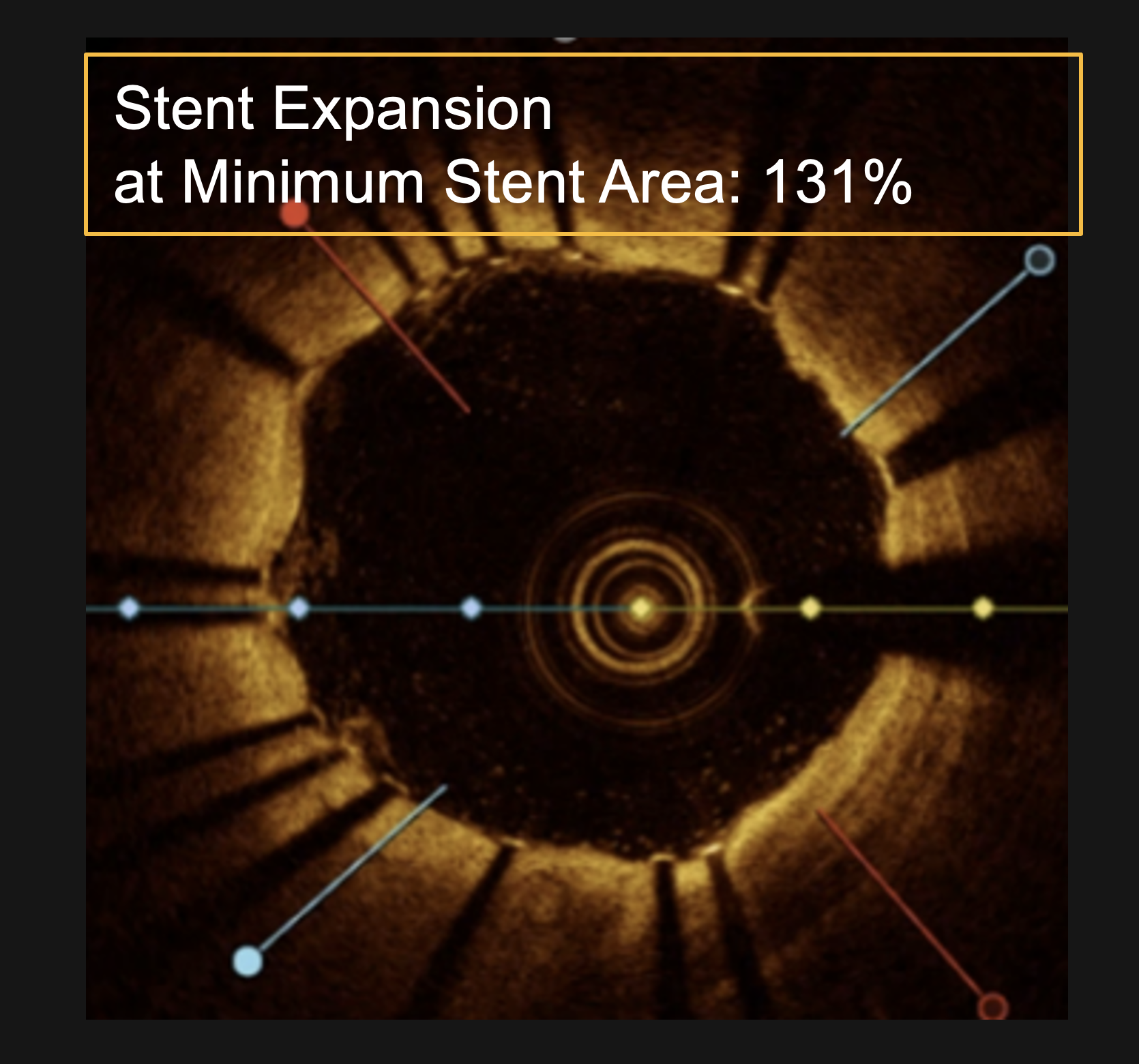
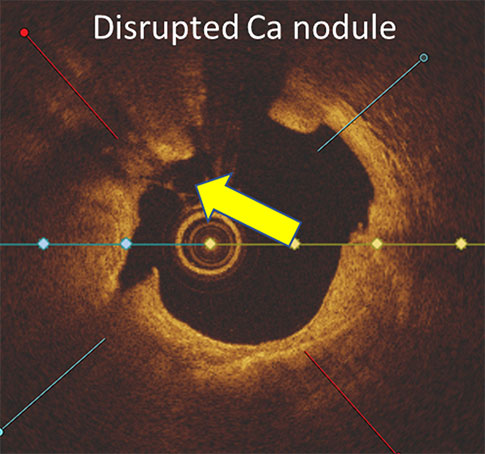
PINNACLE I
(n=60, L=63)
LithiX™ HC-IVL has been proven to successfully treat a wide range of lesion morphologies with 100% angiographic success, no device failure, and very little MACE.
Results demonstrated:
- 98.3% clinical success†
- 100% angiographic success‡
- >100% mean final stent expansion in OCT subset§
†Residual stenosis <30% with no in-hospital MACE through hospital discharge
‡ Stent delivery success, with residual stenosis <50% and no serious angiographic complications
§ in eccentric and concentric calcific lesions and those with calcific nodules
MY-IVL
(n=102, L=130)
A post-market study to evaluate the safety and performance of LithiX™ HC-IVL in a real-world population with highly complex calcified lesions.
The study showed:
- 96.1% freedom from MACE (30 days)
- 96.3% residual stenosis <30% and without intra-procedural MACE
- 113.71% overall mean stent expansion
Study confirms DESyne BDS Plus consistent performance
Elixir Medical’s DESyne BDS Plus system is supported by clinical evidence demonstrating safety and efficacy in drug-eluting stent technology.
DESyne BDS Plus RCT
A prospective, multicenter, single-blind study evaluating the safety and efficacy of DESyne BDS Plus.
Study results:
- Achieved primary endpoint of TLF noninferiority
- No stent thrombosis (definite/probable), CVD, or TV-MI in the DESyne BDS Plus arm
- Sustained low TLF & TVF rates from 12 months to 24 months
Frequently Asked Questions
What clinical evidence supports bioadaptor technology in coronary artery disease?
Bioadaptor technology is supported by peer-reviewed clinical studies demonstrating safety, effectiveness, and long-term outcomes in patients with coronary artery disease. This includes randomized controlled trials and multi-center studies evaluating vessel function, clinical outcomes, and device performance across a range of lesion types.
The clinical studies evaluating the DynamX Coronary Bioadaptor include multiple studies, including the BIOADAPTOR RCT, the INFINITY-SWEDEHEART RCT and the DynamX Mechanistic Clinical Study.
What were the key results of the BIOADAPTOR RCT?
The BIOADAPTOR RCT met its primary endpoint of target lesion failure (TLF) non-inferiority at 12 months compared to a contemporary drug-eluting stent. At 3-year followup, the study shows a flattening of the MACE curve compared to DES.
In addition, the study demonstrated improved early return of vessel function and plaque stabilization. Imaging results using QCA, IVUS, and OCT supported these findings, showing restoration of vessel motion and adaptive behavior.
What clinical outcomes are measured in bioadaptor studies?
Clinical studies of bioadaptor technology evaluate outcomes such as target lesion failure, major adverse cardiac events (MACE), late lumen loss, and percent diameter stenosis.
These endpoints help assess both procedural success and long-term patient outcomes.
In addition to traditional PCI endpoints, DynamX studies also evaluate vessel physiology metrics, including pulsatility and blood flow, vessel motion and adaptive function, adaptive remodeling, and plaque stabilization.
These measurements are designed to assess whether the bioadaptor restores more natural arterial function following PCI.
How does the bioadaptor compare to drug-eluting stents in clinical studies?
Clinical studies comparing the DynamX Coronary Bioadaptor to contemporary drug-eluting stents have demonstrated comparable safety and effectiveness in acute and standard endpoints such as target lesion failure.
In addition, bioadaptor studies evaluate restoration of vessel motion and adaptive function, representing a different approach focused on long-term vessel physiology rather than permanent vessel caging.
Results also showed a novel finding in lipid-rich lesions with plaque regression. Studies showed a statistically significant reduction in TLF at long-term endpoints across all results and in a variety of lesion types and patient populations compared to DES.
Why is randomized controlled trial (RCT) data important in PCI?
Randomized controlled trials are considered a gold standard in clinical research because they reduce bias and allow for direct comparison between treatment approaches. RCT data provide high-quality evidence to support safety and effectiveness in PCI technologies. BIOADAPTOR RCT and INFINITY-SWEDEHEART are 1:1 randomized control trials comparing DynamX bioadaptor to a third-generation drug-eluting stent, the Resolute Onyx.™ All patients in these RCTs are evaluated with follow-up at 6 months, 12 months, and annually through 5 years.
What clinical evidence supports Hertz Contact intravascular lithotripsy (HC-IVL) for treating calcified coronary lesions?
LithiX Hertz Contact intravascular lithotripsy (HC-IVL) is supported by clinical studies demonstrating its effectiveness in modifying calcified coronary lesions and improving vessel expansion during percutaneous coronary intervention (PCI). Evidence includes peer-reviewed data evaluating performance across a range of calcium morphologies, including eccentric, concentric, and nodular lesions.
Clinical outcomes are supported by intravascular imaging, such as optical coherence tomography (OCT) and intravascular ultrasound (IVUS), which have shown consistent plaque fracture and enhanced stent expansion following treatment. Additional evidence includes physician-led case studies and multi-center clinical experience presented at major cardiology conferences, including TCT and EuroPCR.
How does intravascular lithotripsy compare to other calcium modification techniques?
LithiX Hertz Contact intravascular lithotripsy is designed to modify calcified plaque through mechanical energy, enabling vessel expansion while minimizing vessel injury. Compared to other calcium modification techniques, lithotripsy-based approaches demonstrate cracking that can be evaluated using imaging modalities such as OCT and IVUS to confirm plaque fracture and procedural effectiveness.
LithiX HC-IVL studies show fractures in eccentric, concentric, and nodular calcium and optimal stent expansion after treatment.
What evidence supports triple-drug site-specific antithrombotic therapy (TRx) in PCI?
Site-specific antithrombotic therapy (TRx) is supported by clinical research evaluating its potential to address multiple pathways involved in coronary artery disease. This approach combines two anticoagulants, rivaroxaban and argatroban, with an antiproliferative mTOR inhibitor, sirolimus, to target thrombosis and restenosis at the site of disease.
Clinical studies have been designed to assess safety, pharmacologic activity, and effectiveness in reducing thrombotic risk while supporting vessel healing. Evidence includes early clinical investigations and mechanistic studies evaluating drug delivery, local therapeutic effect, and vascular response.
This multi-pathway approach represents a novel strategy in PCI, aiming to improve outcomes by combining antithrombotic and antiproliferative therapies in a single, site-specific treatment.
What types of patients are included in clinical studies of PCI technologies?
Clinical studies typically include patients with a range of coronary artery disease presentations, including complex lesions, calcified vessels, and high-risk patient populations. This helps evaluate device performance across real-world patient populations.
PMN 1710 Rev H

