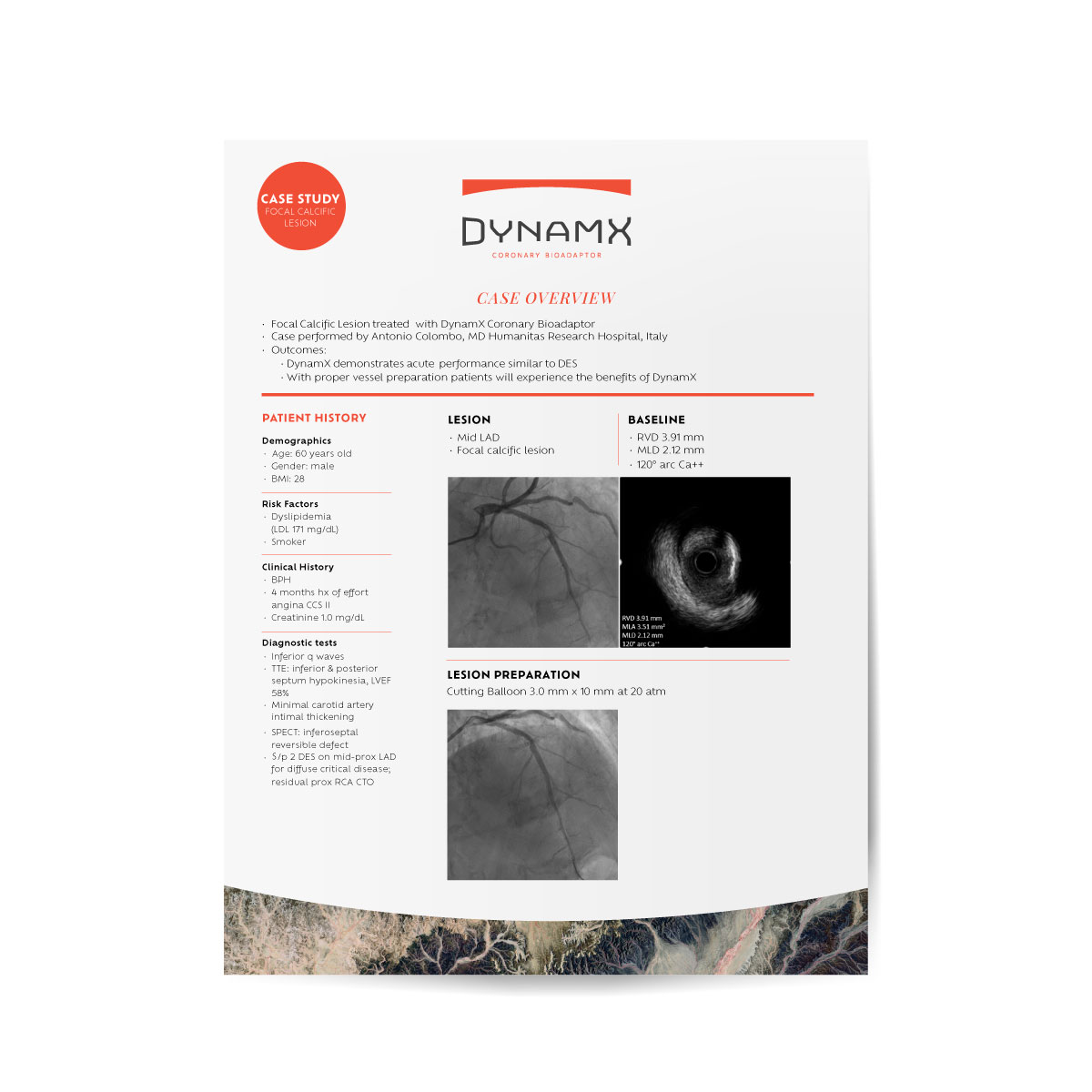
CASE STUDIES
& VIDEOS
CASE STUDIES
& VIDEOS
Hear from leaders in interventional cardiology as they discuss our unique technologies and clinical evidence. Or take a deep dive into how a technology works.





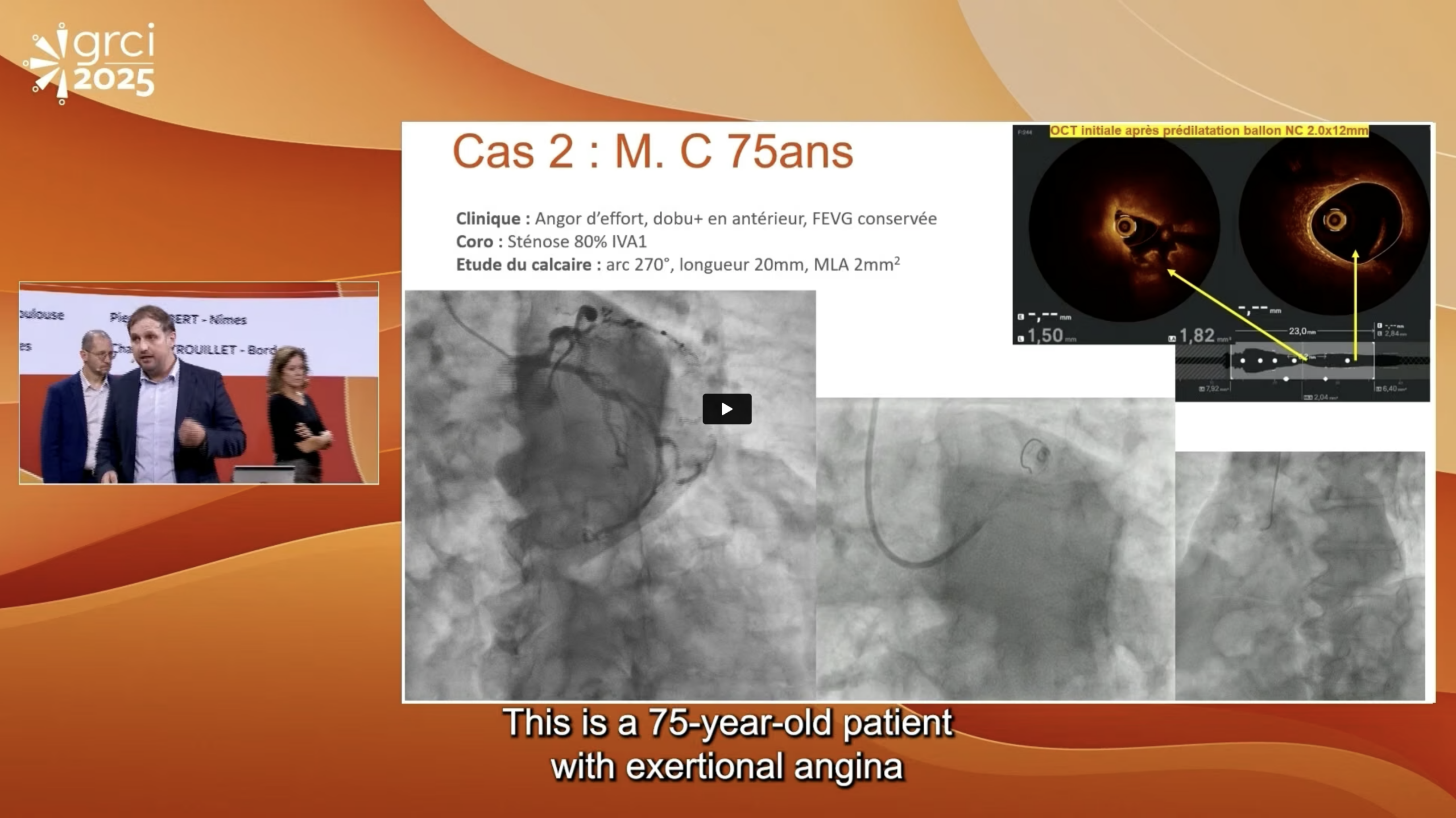
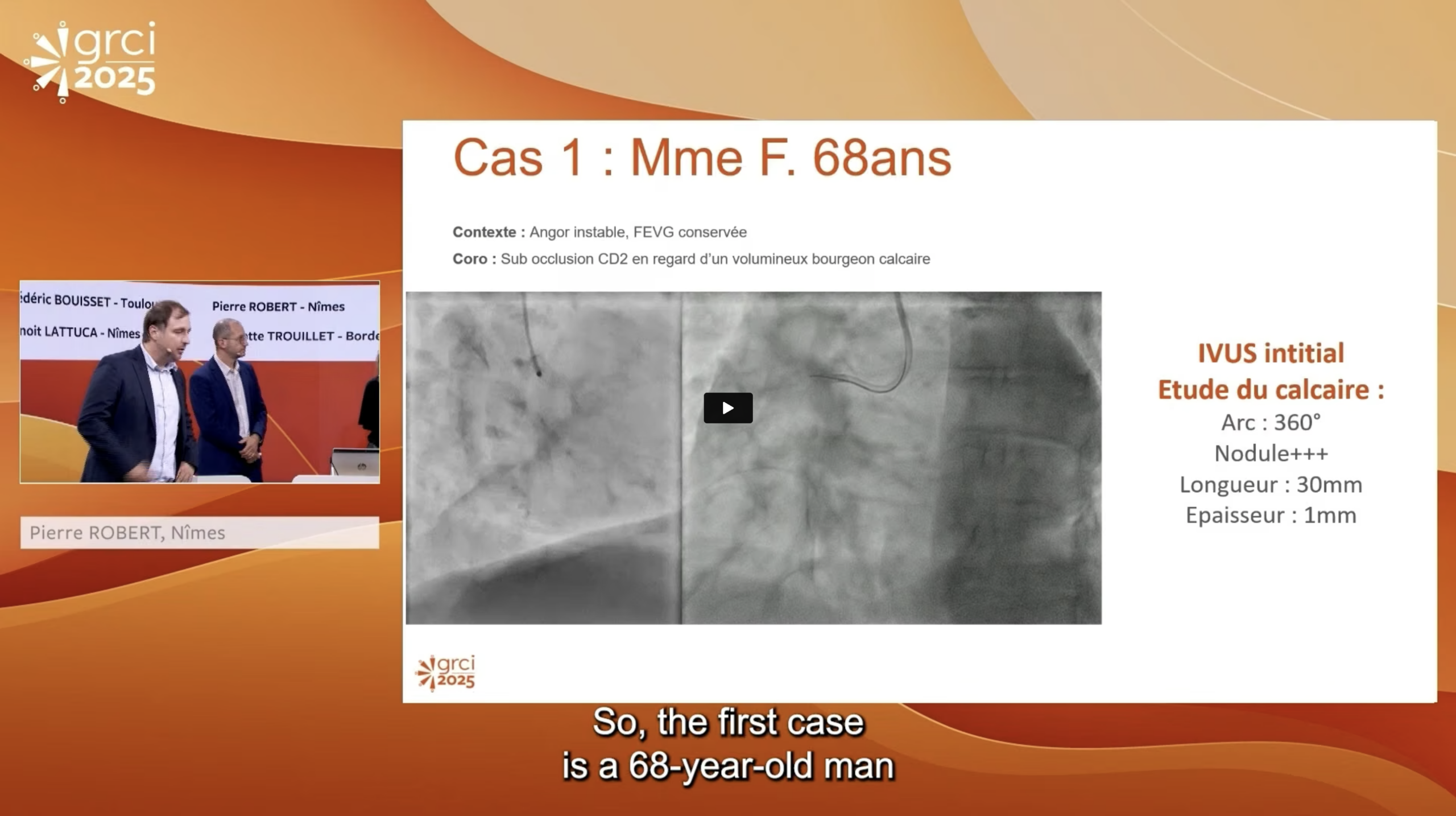
Interview with Dr. Shigeru Saito: BIOADAPTOR RCT results comparing novel DynamX Bioadaptor to Resolute Onyx DES. Dr. Saito explains the long-term risks associated with drug-eluting stents and the limitations of DES design. He shares key findings of the trial and how the unique design of the bioadaptor contributes to the significantly superior outcomes.
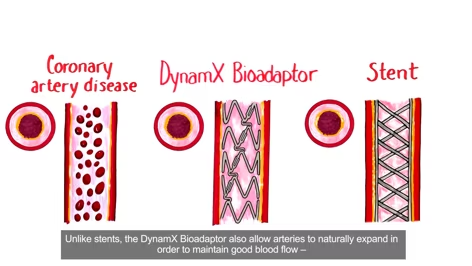
What is Coronary Artery Disease and Why Treating it with a Bioadaptor Matters? Learn how people are affected by coronary artery disease and its treatments. Understand disease treatment with stents and how they restore blood flow but restrict natural vessel motion and function, leading to major health issues. The DynamX coronary bioadaptor is a significant innovation that not only restores blood flow but also releases the vessel and allows it to pulsate more freely, increasing vital blood flow and returning it to a more natural state.
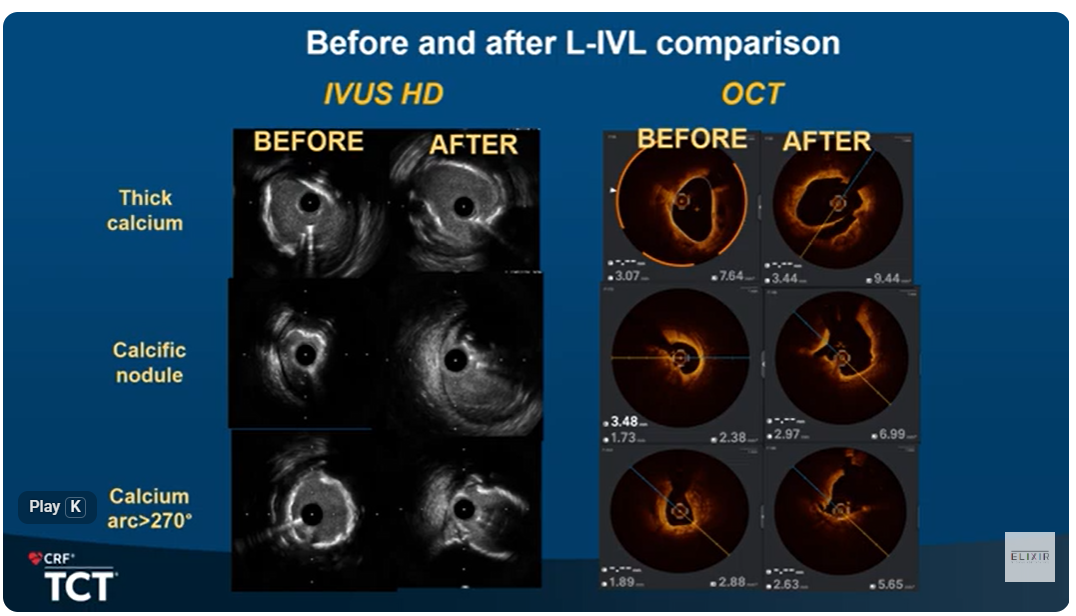
Interview with Dr. Stefan Verheye: BIOADAPTOR RCT results comparing novel DynamX Bioadaptor to Resolute Onyx DES. Dr. Verheye explains how the bioadaptor allows for restoration of vessel motion and function unlike the comparative DES. Pulsatility is restored resulting in significantly increased blood flow and lumen volume increased confirming adaptive remodeling when the vessel is uncaged. Findings also suggest potential benefits in plaque stabilization and regression.
The Future of Vascular Intervention. Working with the vessel to restore and increase blood flow, releasing and uncaging the vessel to move freely and returning the vessel to its natural physiology.
In this webinar on the treatment of coronary artery disease leading experts are exploring the latest clinical advancements in PCI and specifically how DynamX Bioadaptor could further improve PCI outcomes over DES. This session is featuring an insightful case discussions and recent clinical evidence on DynamX Bioadaptor. INFINITY-SWEDEHEART study outcomes published in Lancet demonstrated significant improvements of DynamX Bioadaptor over contemporary DES. Learn about the clinical evidence on DynamX.
The opinions expressed here belong solely to the physician and do not reflect the views of Elixir Medical Corporation. DynamX, Elixir are registered trademarks in the U.S. and internationally for Elixir Medical Corporation. DynamX Coronary Bioadaptor System is CE Marked. Not approved in the U.S. 2024 Elixir Medical. All rights reserved. International (OUS) Use Only. Indications, contraindications, warnings and instructions for use can be found in the product labeling supplied with each device.
Slide at the time period 00:08:16 – 00:08:54 has an error. The correct number of patients is n=48 for DynamX and n=47 for Onyx. The standard deviation for the flow volume increase is 8.9 for DynamX and 8.0 for Onyx. Slide at the time period 00:12:55 – 00:13:40 has an error. The correct P value is “Long P-value = 0.0079”. Slide at the time period 00:14:02 – 00:14:13 has an error. The correct P value is “Long P-value = 0.011”
PMN 2116 Rev A
PMN 1711 Rev B